Open 24/7
Advanced and compassionate care for your pet
At Maine Veterinary Medical Center in Scarborough, ME, we offer comprehensive veterinary services to meet the needs of your pet.
Request your consultation today!

We’ve taken vet care to the next level.
Maine Veterinary Medical Center is proud to be one of 26 hospitals in the United States operating as a Certified Level 1 Facility with the Veterinary Emergency and Critical Care Society. With state-of-the-art facilities and cutting-edge technology, our veterinarians can deliver diagnostic services and advanced treatment options for a wide range of conditions.

We specialize in excellence.

We’re proud to be a Level 1 VECCS facility.
The Veterinary Emergency and Critical Care Society (VECCS) offers certification programs to veterinary facilities that provide emergency and critical patient care. The VECCS does this to raise the standard of care while also increasing public and professional awareness in the area of veterinary emergency and critical patient care.

Complete Veterinary Care
At Maine Veterinary
Maine Veterinary Medical Center is committed to providing high-quality care for pets of all ages and needs.
Our veterinary team offers specialized veterinary services in internal medicine, neurology, oncology, ophthalmology, surgery, sports medicine and rehabilitation, emergency and critical care, and cardiology.
Veterinary Surgery
The surgical department at MVMC is dedicated to providing excellence in small animals…
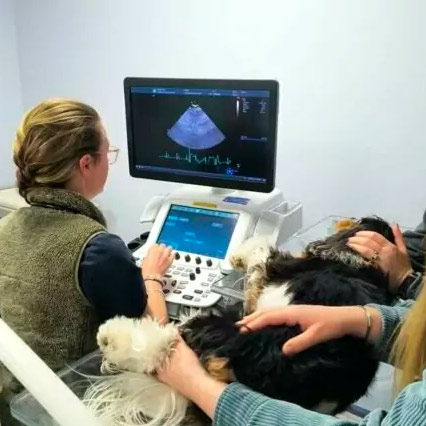
Veterinary Cardiology
Our cardiology team is dedicated to diagnosing, managing, and treating disorders of the heart and cardiovascular systems.
Veterinary Neurology
We are on your side.
Meet our veterinary team.
The veterinary team at Maine Veterinary Medical Center consists of highly trained and knowledgeable problem solvers. Our professional staff members and technicians who work alongside our board-certified veterinarians are lifelong animal lovers on a mission to enhance pet quality of life.
Kind words from clients.
“Dr. Gill and her team were truly wonderful during the time my dog, Leo, was a patient at MVMC. They showed incredible patience in answering all of my questions (sometimes multiple phone calls a day) as well as in taking the time to carefully explain each new treatment/medication/whatever came up throughout the process. I will be forever grateful for the amazing care that Leo received.”
“The doctors, technicians, and staff here are wonderful. We have been for multiple reasons (PT, acupuncture, emergency services), and every single person has been kind, understanding, knowledgeable, and professional. I appreciate that they are very willing to take the time to explain treatment and cost options to figure out what works best for our needs.”
Aimee H.